Charles Law
The volume of a fixed mass of gas is directly proportional to its temperature at constant pressure.
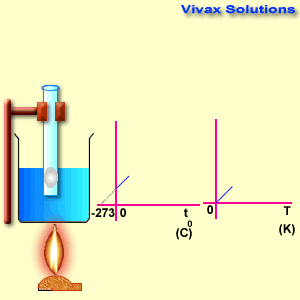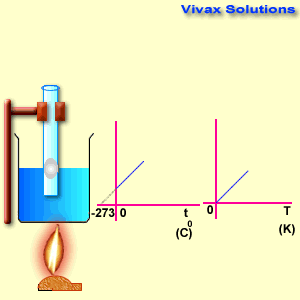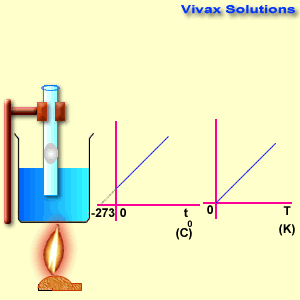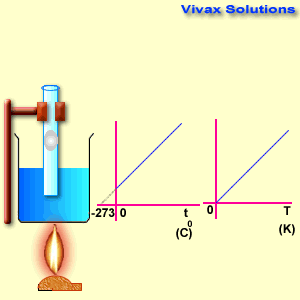
Please move the slider and experiment with it.
V ∝ T
v ∝ t
v = k t
v = k t
As you can see, the line, when extended backwards crosses the x-axis at -273 C0. There is a special temperature when all gases have zero volume, called Absolute Temperature.
So, 0 T = -273 C
+ 273 => 273 T = 0C
+ t => (273 + t) T = t C
T = 273 + t, where t is the temperature in Celsius.
If the temperature and volume of a fixed mass of gas take the values of v1 T1and V2 T2 respectively, where T is absolute
temperature,
V1 / T1 = V2 / T2
E.g.
The volume of a fixed mass of air is volume is 8 cm3 at 27 C0. Its
temperature is raised to 127 C0, while keeping the pressure constant. Find the volume.
p
1 = 20, v
1 = 8, p
2 = 40
According to Charles's law,
V
1 / T
1 = V
2 / T
2
8 / (273 + 27) = v
2 / (273 + 127)
v
2 = 10.6cm
3
Please answer the following questions.
- The volume of a gas at 30C0 is 8 cm3. Find its volume when the
temperature is 50C0 while pressure remains the same.
- The volume of a gas at constant temperature is plotted against temperature in Celsius. What do the gradient and intercept
represent ?
- The volume of a gas at 30C0 is x cm3. Find x when the
temperature is 50C0 while pressure remains the same.